Neuland Laboratories is a leading, global active pharmaceutical ingredient (API) manufacturing and development organization that caters to the pharmaceutical and biotech industry’s chemistry needs. Right from synthesis of pre-clinical compounds to supplying New Chemical Entities (NCEs) and advanced intermediates at various stages in the clinical life-cycle, as well as commercial & generics, we offer agile and flexible API manufacturing and development services.
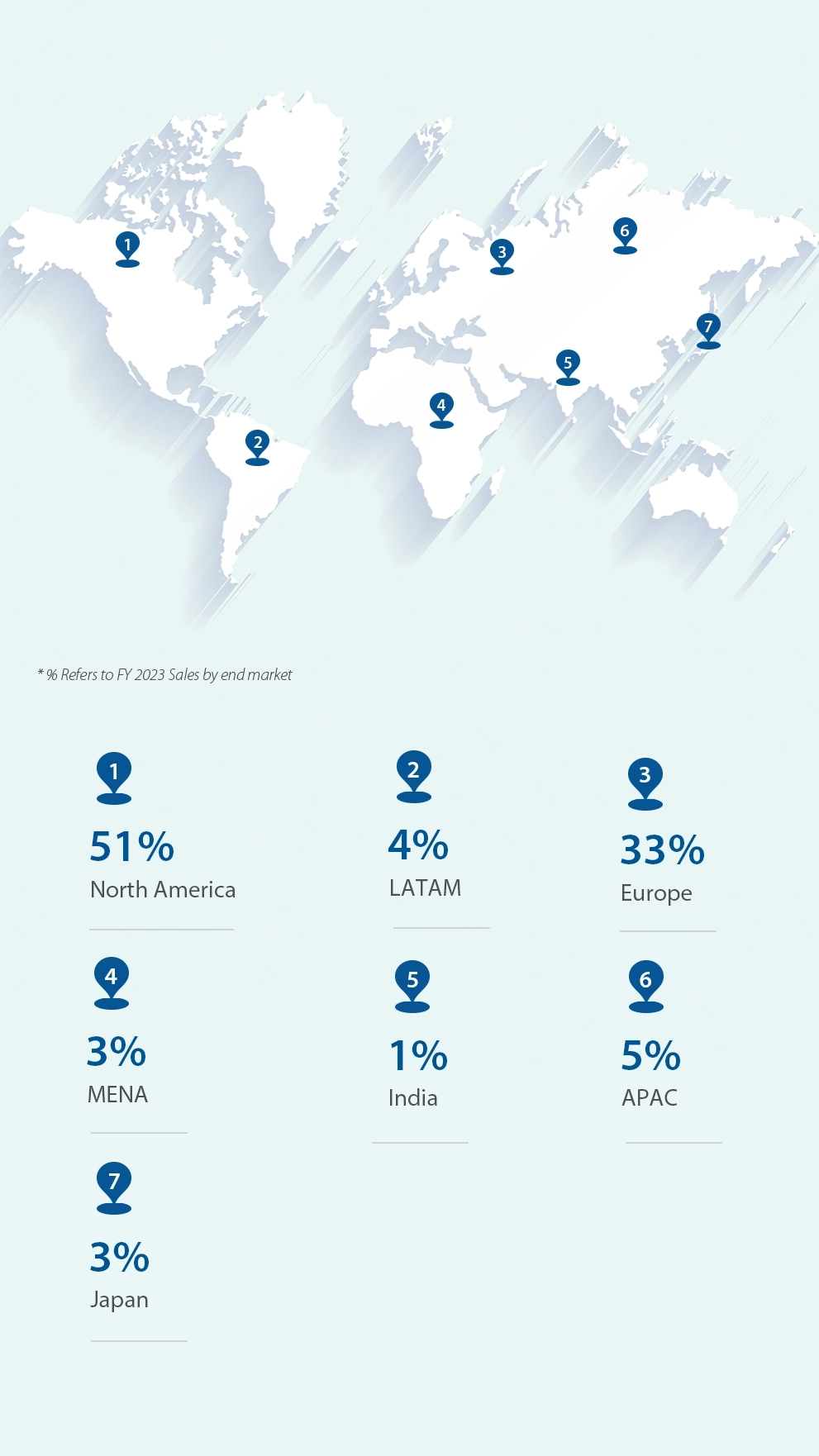
We cater to over 500 customers in 80+ countries. Our complex chemistry capabilities together with our regulatory-compliant manufacturing facilities have made us a trusted provider of APIs and advanced intermediates. The Company’s product portfolio includes over 100 APIs across 10 diverse therapeutic areas.
Our custom development services include a full range of the pharmaceutical industry’s chemistry requirements, right from pre-IND through commercial manufacturing. We offer both small-scale clinical trial quantities and full commercial-scale supply with minimal tech transfer timelines. Neuland’s peptide synthesis services include production of peptides from milligrams to multi-kilogram scale by standard sequential chemical peptide syntheses and segment condensation strategies. Neuland has expertise in both solution phase, solid phase synthesis and hybrid methodologies.
At Neuland, we are actively integrating sustainability into our business by taking cognizance of stakeholder expectations across environmental, social and governance (ESG) parameters. The spectrum ranges from efficient usage of chemicals and solvents, climate protection, ethical business practices, sustainable supply chain, the health and safety of our people to robust risk management along the value chain.
We are headquartered in Hyderabad, India and our manufacturing and research facilities are situated near Hyderabad. The Company has Business development offices in the US, Europe and Japan.

Our Values
We follow a strong set of values, termed ‘The Neuland Way’, which spurs integrity and motivation among the workforce.
Neuland in Numbers
39+
years of experience
907 KL
API manufacturing capacity
100+
APIs across 10 therapeutic areas
80+
countries where customers are located
950+
Drug Master Files (DMFs) worldwide
65
active US DMFs
73%
revenue from exports
1,573
employees
₹1,200.9 crores
Total Income
₹281.1 crores
₹281.1 crores
₹163.1 crores
Profit after Tax
₹953.2 crores
FY 2022
₹144.3 crores
FY 2022
₹63.5 crores
FY 2022
Silver Sustainability rating
given by ECOVADIS FY 2022

3,000+
saplings planted
85%
employees covered under leadership & development interventions
92%
treated wastewater recycled
15%
reduction in carbon usage
Our Revenue by Geography